Blog
DMARD monitoring during the COVID-19 pandemic
We describe some of our recent work investigating the impact of the COVID-19 pandemic on DMARD safety monitoring across >24 million patients’ records in England.
Read more about DMARD monitoring during the COVID-19 pandemic →
OpenSAFELY research software advocate vacancy
We’re hiring a research software advocate and we think you’d be a good fit
Read more about OpenSAFELY research software advocate vacancy →
OpenSAFELY software engineering vacancies
We’re hiring software people and we’d love you to join us
Read more about OpenSAFELY software engineering vacancies →
The impact of COVID-19 on medication reviews in English primary care. An OpenSAFELY analysis. Part 2: Results
In the second of a two-part blog series, pharmacists Chris Wood and Vicky Speed talk about the results of their research on medication review activity
Read more about The impact of COVID-19 on medication reviews in English primary care. An OpenSAFELY analysis. Part 2: Results →
The impact of COVID-19 on medication reviews in English primary care. An OpenSAFELY analysis. Part 1: Analysis Design
In the first of a two-part blog series, pharmacists Chris Wood and Vicky Speed talk about designing their research on medication review activity
Read more about The impact of COVID-19 on medication reviews in English primary care. An OpenSAFELY analysis. Part 1: Analysis Design →
Improvement Radar: A new OpenPrescribing tool to identify best practice
We’ve launched a new OpenPrescribing tool, the Improvement Radar, which makes it easy to identify organisations which have shown substantial improvement.
Read more about Improvement Radar: A new OpenPrescribing tool to identify best practice →

The OpenSAFELY tech teams' work pipeline
How we organize and plan work for our tech teams
Read more about The OpenSAFELY tech teams' work pipeline →
Can I do a Doctorate at the Bennett Institute?
Are you interested in doing a DPhil at the Bennett Institute? This post contains details on how to do so!.
Read more about Can I do a Doctorate at the Bennett Institute? →
Ben Goldacre appears on BBC's More or Less
Ben discusses the OpenSAFELY platform with fellow nerd Tim Harford
Read more about Ben Goldacre appears on BBC's More or Less →
STAR-PUs - should we still be using them?
In this blog we explain some of the issues with using STAR-PUs and ask for your input on how we should use them in future.
Read more about STAR-PUs - should we still be using them? →
OpenPrescribing Winter 2023/2024 Newsletter
This newsletter contains all the latest updates from OpenPrescribing
Read more about OpenPrescribing Winter 2023/2024 Newsletter →
Research Integrity Update - Feb 2024
Details on two new papers from the Bennett Institute Research Integrity Team.
Read more about Research Integrity Update - Feb 2024 →
PINCER: Monitoring medication safety throughout the COVID-19 pandemic using OpenSAFELY
Here we describe some of our recent work investigating the impact of the COVID-19 pandemic on safe prescribing using a set of quality assured indicators across 57 million patients’ records in England
Read more about PINCER: Monitoring medication safety throughout the COVID-19 pandemic using OpenSAFELY →
Spot the difference. Part 2: Analytic Choices
In the second of a two-part blog we explain why OpenPrescribing figures don’t always match with other prescribing data resources, due to differences in how data is presented.
Read more about Spot the difference. Part 2: Analytic Choices →
Spot the difference. Part 1: Source Datasets
In the first of a two-part blog we explain why OpenPrescribing figures don’t always match with other prescribing data resources, due to differences in the data source.
Read more about Spot the difference. Part 1: Source Datasets →
Trends in inequalities in avoidable hospitalisations across the COVID-19 pandemic
In this guest blog, Mark Green describes their latest paper using OpenSAFELY.
Read more about Trends in inequalities in avoidable hospitalisations across the COVID-19 pandemic →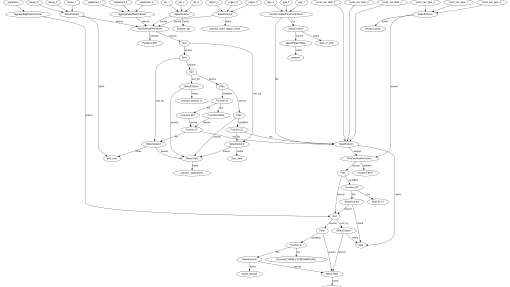
How we test ehrQL
How can you be confident that the queries you write will be correctly interpreted and return the expected results?
Read more about How we test ehrQL →
